
International Journal of Neuropsychopharmacology (2020) XX(XX): 1–16; pyaa075, https://doi.org/10.1093/ijnp/pyaa075
Anti-Amnesic and Neuroprotective Effects of Fluoroethylnormemantine in a Pharmacological Mouse Model of Alzheimer’s Disease. Simon Couly, Morgane Denus, Mélanie Bouchet, Gilles Rubinstenn, Tangui Maurice.
Background
Current therapies in Alzheimer’s disease (AD), including Memantine, have proven to be only symptomatic but not curative or disease modifying. Fluoroethylnormemantine (FENM) is a structural analogue of Memantine, functionalized with a fluorine group that allowed its use as a positron emission tomography tracer. We here analyzed FENM neuroprotective potential in a pharmacological model of AD compared with Memantine.
Methods
Swiss mice were treated intracerebroventricularly with aggregated Aβ 25–35 peptide and examined after 1 week in a battery of memory tests (spontaneous alternation, passive avoidance, object recognition, place learning in the water-maze, topographic memory in the Hamlet). Toxicity induced in the mouse hippocampus or cortex was analyzed biochemically or morphologically.
Results
Both Memantine and FENM showed symptomatic anti-amnesic effects in Aβ 25-35-treated mice. Interestingly, FENM was not amnesic when tested alone at 10 mg/kg, contrarily to Memantine. Drugs injected once per day prevented Aβ 25-35-induced memory deficits, oxidative stress (lipid peroxidation, cytochrome c release), inflammation (interleukin-6, tumor necrosis factor-α increases; glial fibrillary acidic protein and Iba1 immunoreactivity in the hippocampus and cortex), and apoptosis and cell loss (Bcl-2–associated X/B-cell lymphoma 2 ratio; cell loss in the hippocampus CA1 area). However, FENM effects were more robust than observed with Memantine, with significant attenuations vs the Aβ 25-35-treated group.
Conclusions
FENM therefore appeared as a potent neuroprotective drug in an AD model, with a superior efficacy compared with Memantine and an absence of direct amnesic effect at higher doses. These results open the possibility to use the compound at more relevant dosages than those actually proposed in Memantine treatment for AD.
Read the full paper
Preview of the work:
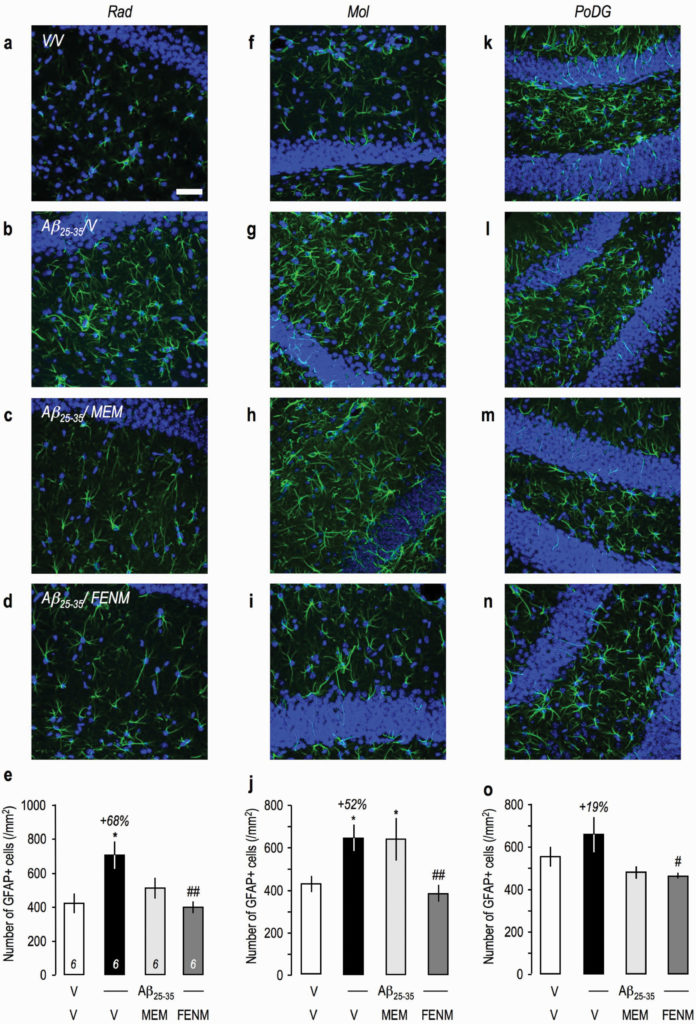
Protective effects of Memantine and FENM administered at 0.3 mg/kg IP on the astroglial reaction in the hippocampus of Aβ 25-35-treated mice using GFAP immunolabeling: (a–e) stratum radiatum, (f–j) molecular layer, and (k–o) polymorph layer with (a–d, f–i, k–n) typical immunofluorescence micrographs (blue: DAPI, green: GFAP) and (e, j, o) quantifications. Coronal 25-µm-thick sections were stained with anti-GFAP antibody and 3 areas of the hippocampus analyzed as shown in Figure 8g. Scale bar (a) = 50 µm. ANOVA: F(3,22) = 5.06, P < .01 (e); F(3,23) = 4.50, P < .05 (j); F(3,23) = 3.24, P < .05 (o). *P < .05, ***P < .001 vs the (V+V)-treated group; #P < .05, ##P < .01 vs the (Aβ 25–35+V)-treated group; Dunnett’s test.
